
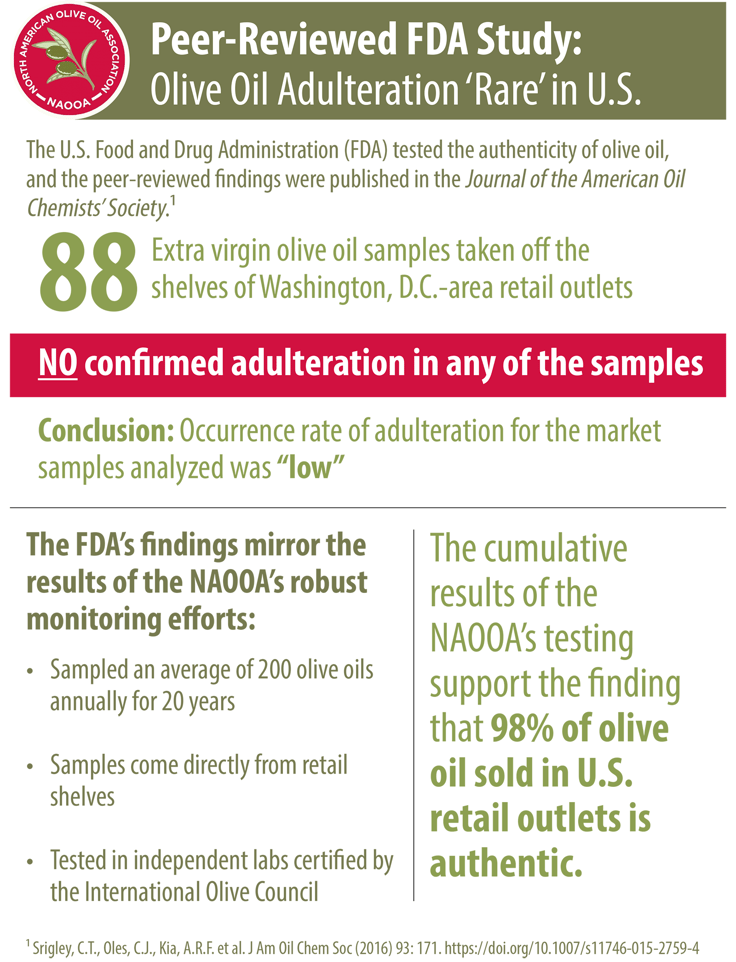
The FDA conducted an independent study that tested 88 different olive oils in the United States and turned up no evidence of adulteration.
About the FDA's Study
Researchers purchased 88 samples of extra virgin olive oil off the shelves from Washington, D.C. area supermarkets, and published its results in the Journal of the American Oil Chemist's Society with the title "Authenticity Assessment of Extra Virgin Olive Oil: Evaluation of Desmethylsterols and Triterpene Dialcohols" in December of 2015.
The study indicated that olive oil adulteration is rare in the United States. While three of the 88 samples failed to meet some of the purity criteria (which would translate to about a 3.4% failure rate), because those deviations could reasonably have been explained by the fact the oils in question came from geographic areas known for legitimate deviations, even those three samples could not be conclusively be said to be adulterated. As a result, the FDA concluded:
"Overall, a low occurrence rate of adulteration (<5 %) was found for market samples of EVOO based on purity criteria for total sterol content, desmethylsterol composition, and content of triterpene dialcohols, as specified in the US Standards for grades of olive oil and olive-pomace oil."
To further assure consumers, the NAOOA manages a Quality Seal Program. Seal brands are tested twice per year and are certified to meet International Olive Council standards.
Download our infographic on the FDA's findings as a PDF version.






